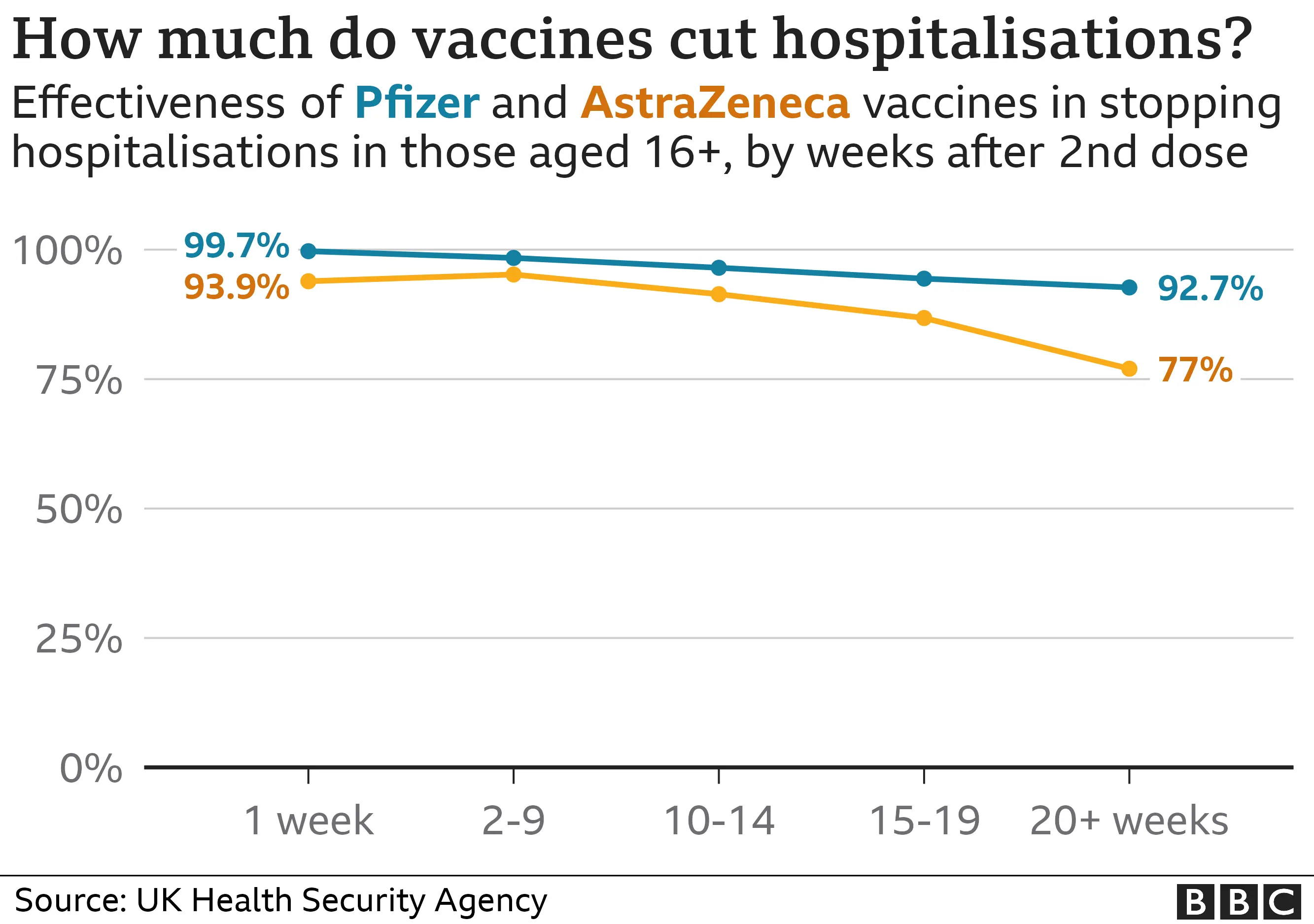
The Centers for Disease Control and Prevention blocked publication of a study showing covid-19 vaccines reduced emergency department visits and hospitalizations by about half among healthy adults last winter, according to three people familiar with the decision who spoke anonymously due to fear of retaliation.
The report was previously delayed by the CDC director
The study had already faced an earlier delay initiated by the head of the Centers for Disease Control and Prevention before being blocked from appearing in the agency’s flagship scientific journal, the sources said.
Vaccine effectiveness was measured in real-world conditions
The blocked analysis found that covid-19 shots cut the likelihood of hospital-related care by approximately 50 percent during the most recent winter season, based on data from healthy adult populations.
Internal disagreement over release persists
Three individuals with knowledge of the internal process described a split within the agency over whether to publish the findings, with some officials advocating for transparency and others opposing release.

Why did the CDC block the report?
The sources did not specify the exact reasons for blocking the study, only that the decision was made despite prior delays and involved concerns among agency staff about potential repercussions for speaking out.
What does the blocked data suggest about vaccine protection?
The data indicates that covid-19 vaccines provided substantial protection against severe outcomes requiring emergency or hospital care in healthy adults during the past winter, reducing such visits by roughly half.